 Skip to content
Skip to content


At NST Law, we are dedicated to helping injured individuals and their families get the justice and compensation they deserve.

Millions of people rely on devices manufactured by Philips Respironics to control medical conditions such as sleep apnea. Koninklijke Philips N.V., better known as Philips, is a global corporation headquartered in Amsterdam. Philips and its American subsidiary, Philips North America, manufacture products in numerous industries, including consumer goods, electronics, and healthcare.
On April 26, 2021, Philips announced potential health risks associated with certain products within its Sleep & Respiratory Care vision, specifically Continuous Positive Airway Pressure (CPAP) machines, Bi-Level Positive Airway Pressure (BiPAP) machines, and ventilators. On June 14, 2021, Philips formally issued a recall notice for specific devices manufactured between 2009 and April 26, 2021. Philips refers to itself as a “global leader in Sleep Diagnostic and Therapy Solutions” and is the largest manufacturer of CPAP devices in the world.
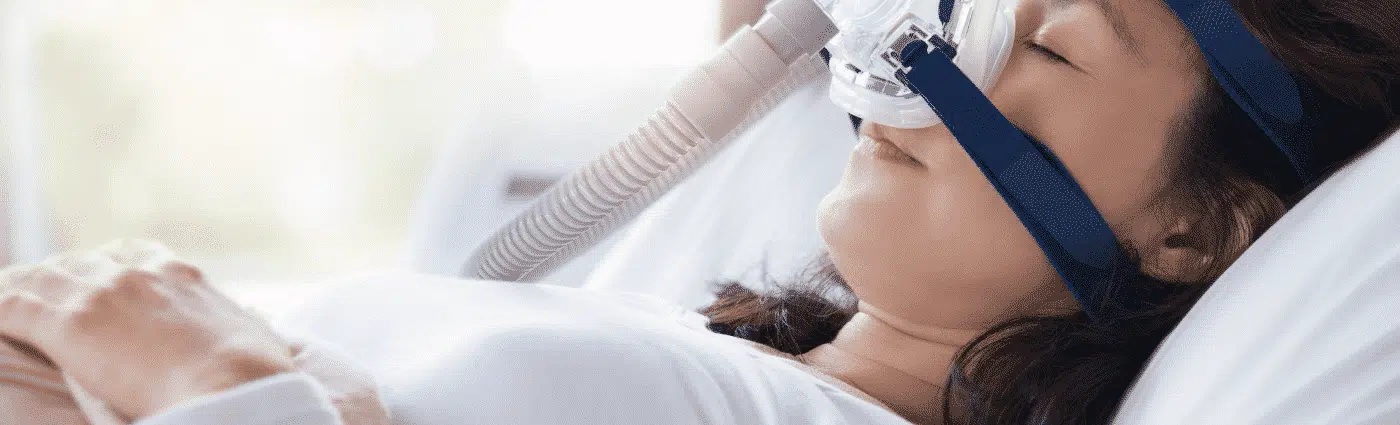
Sleep Apnea is a serious medical condition involving abnormal breathing during sleep, where breathing stops and starts. Lapses in breathing during sleep can negatively affect the body’s oxygen supply. There are three types of sleep apnea:
Symptoms of sleep apnea can include heavy snoring, gasping for air during sleep, insomnia, waking up with a dry mouth, and stopping breathing during sleep. If you experience any of these symptoms, you should consult with your doctor immediately. A common treatment for sleep apnea is your doctor prescribing a CPAP or BiPAP machine to use on a nightly basis.
On April 26, 2021, Philips announced potential health risks associated with their CPAP, BiPAP, and ventilator devices distributed between 2009 and April 26, 2021. Philips formally issued a recall notice for these devices on June 14, 2021. On June 30, 2021, the FDA issued an alert that certain Philips ventilators, CPAP, and BiPAP machines were recalled by Philips due to “potential health risks,” specifically that foam designed to reduce sound and vibration can break down and enter the device’s air pathway. On July 23, 2021, the FDA classified this recall as a Class 1 recall, the most serious classification available. A Class 1 recall means the use of that device could cause serious injury or death. If you were prescribed and used a CPAP, BiPAP, or ventilator device manufactured by Philips, you should consult with your doctor, as use of the device may cause medical issues. However, you should not stop using a prescribed device before consulting with a physician.
The recalled Philips devices contain polyester-based polyurethane (PE-PUR) sound abatement foam which is used to reduce sound and vibration in the devices. In these devices, the foam may break down and enter the air pathway of the device. If this happens, chemicals or black debris from the foam could be inhaled or swallowed by the user of the device. Exposure to the black debris or chemicals may cause serious injuries, including irritation, inflammation, headaches, asthma, adverse effects to organs (including liver and kidney), and even cancer. It is alleged that Philips knew of these dangers yet failed to take action or properly warn consumers.

To date, Philips has issued a recall for the following CPAP and BiPAP devices distributed from 2009 through April 26, 2021:
Philips issued a recall for the following ventilators distributed from 2009 through April 26, 2021:
Our medical device lawyers are currently investigating links between Philips CPAP, BiPAP, and ventilator machines and injuries such as cancer. The following injuries may be linked to inhaling polyurethane foam from an affected Philips device:
If you use a Philips CPAP machine, BiPAP machine, APAP machine, or ventilator and have experienced any of the above symptoms or been diagnosed with any of these conditions, you should consult your doctor immediately. Philips has sent users of affected devices a letter explaining the device is subject to a recall and that further use could cause risk of serious illness, disease, or death. Our Philips CPAP recall lawyers are available to speak with you about your legal rights and options.
Nationwide, lawsuits are being filed by injured victims against Philips and Philips North America for injuries caused by the recalled devices. The lawsuits allege Philips has known about the risks of their sound abatement foam for years but failed to properly warn consumers or the public. Further, lawsuits filed by attorneys representing victims of CPAP machines have alleged Philips negligently and defectively designed their products, knowingly sold defective devices harmful to consumers, and failed to warn consumers of the risks of polyester-based polyurethane (PE-PUR) sound abatement foam. Under product liability laws, a medical device manufacturer can be held liable for the negligent design of a device, and where side effects are either known or should be known but not disclosed, negligently failing to warn consumers and patients.
Users of the Philips machines affected by the degradation of the foam have reported experiencing pain and discomfort, which could entitle them to monetary damages. Additionally, these individuals have other medical conditions that have been worsened by the continued use of the CPAP machines. The possibility of suffering further harm due to a product that was supposed to help can be devastating. An experienced lawyer can assess your situation, explain your legal rights, and help you pursue justice. Those injured by a Philips CPAP machine may be able to recover compensation for medical bills, lost wages, pain and suffering, and more. If the Philips device caused a loved one to pass away, a wrongful death lawsuit may be filed.

If you or your loved one used a Philips CPAP, BiPAP or mechanical ventilator machine and has been affected by the recall, you may be eligible to recover compensation for medical bills, lost wages, pain and suffering, and other damages. For a free initial consultation with a CPAP injury lawyer today, call NST Law at 800-529-4004 or contact us online. NST Law represents people nationwide in product liability, mass tort, medical device, and pharmaceutical cases, and our team has recovered over $1.5 billion in compensation for clients over the last 30+ years.
Memphis Office
488 S. Mendenhall Road
Memphis, TN 38117
Phone: 901-683-7000
Knoxville Office
408 N. Cedar Bluff Road Suite 450
Knoxville, TN 37923
Phone: 865-684-1000
Jackson, TN Office
2709 N. Highland Avenue
Jackson, TN 38305
Phone: 731-427-5550
Tupelo Office
201 Magazine Street Suite 201
Tupelo, MS 38804
Phone: 662-856-1000
Jackson, MS Office
125 S Congress St Suite 1300,
Jackson, MS 39201
Phone: 601-488-3483
Hernando, MS Office
2475 Memphis Street,
Hernando, MS 38632
Phone: 662-350-9100
Jonesboro Office
317 Southwest Drive Suite B
Jonesboro, AR 72401
Phone: 870-931-0500
Little Rock Office
4 Shackleford Plaza Suite 211
Little Rock, AR 72211
Phone: 501-435-0000
St. Louis Office
701 Market St Suite 285
St. Louis, MO 63101
Phone: 314-476-0500
Cape Girardeau Office
1918 N. Kingshighway Suite 102
Cape Girardeau, MO 63701
Phone: 573-262-9311
Belleville Office
6400 W Main St. Suite 10
Belleville, IL 62223
Phone: 618-215-5631